|
Happy New Year!! Every student got a review packet today. They completed the first and second page of the packet in class. They will be taking a quiz everyday to get extra credits on the MAJOR grade. If absent, please study over Unit 2- matter by using attached review packet and be ready to take the quiz tomorrow in class.
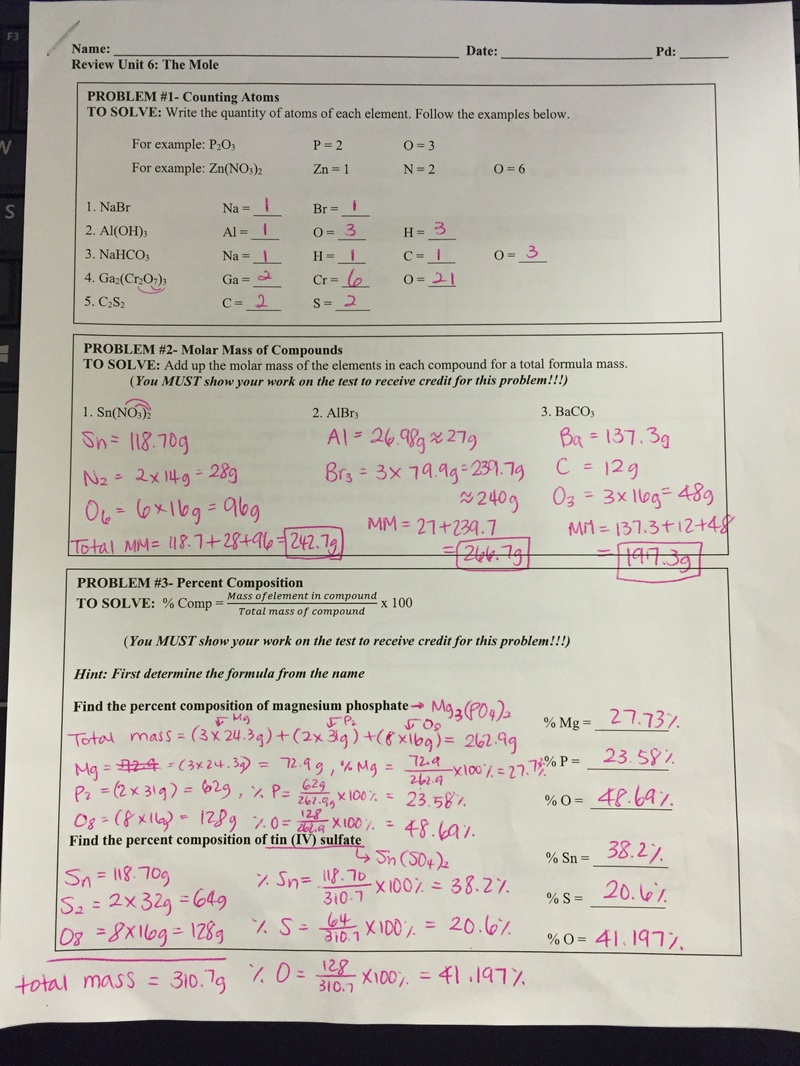
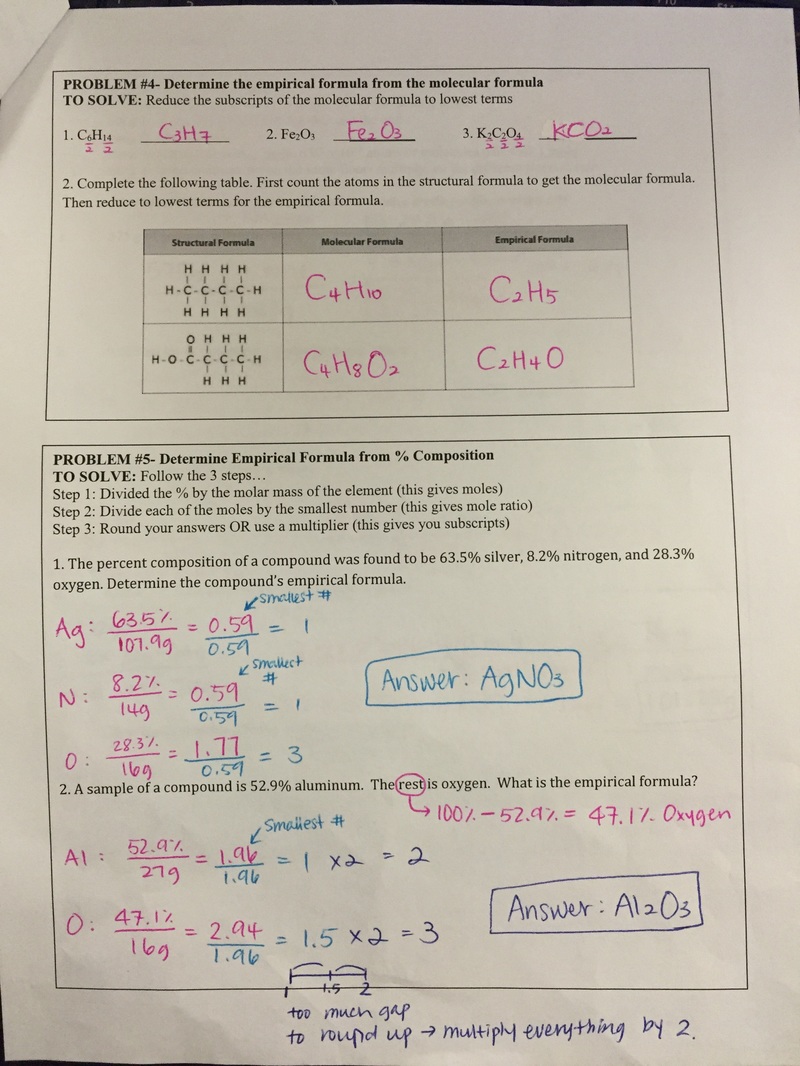
Students took practice exam today. Below is the questions and answers. The actual exam will have exactly same format as the practice exam but formulas and numbers will be different. There will not be word banks provided on the actual test.
If absent, study below questions and prepare well for the exam tomorrow. Students learned how to convert moles to grams and grams to mole of an element and compounds. The conversion steps are simple; 1. Write the given number down. 2. Draw a cross. 3. grams (g) is always pairing with '1 mole'. 4. If the numbers are on the same floor, you multiply them. If the numbers are on the different floor, you divide them. If absent, watch the following video and ask Ms. Ha for the notes and worksheets. They are two minor grades and due by Friday (1/2/17). Students could find important facts about mole and be able to calculate moles to grams. If absent, read the ppt carefully and complete the worksheet.
Students were conducting bubble gum lab to calculate percent composition of sugars in a gum.
If absent, please come to Ms. Ha to get the lab worksheet. Please watch the following video and correct the test questions. To correct them, you must use a separate sheet of paper and put: Question number l your first answer l right answer l why is this right answer? The explanation part (why is this right answer?) is the most important. Without the correct and detail explanation, there will be no credit for the correction. Here are the video you should watch before the correction:
Students worked on a worksheet to practice over the percent composition. Students were able to get a clearer concept and skills to calculate the mass % composition of atoms in chemical formula. If absent, please watch the following video, complete the worksheet and turn it in to Ms. Ha before the Thanks Giving break. Also please make sure that you will be retaking the Unit 5 test tomorrow (Friday, 11/18/16)
Today students learned about percent composition of atoms in chemical formula. They played an activity finding percent composition of ingredients of hamburger. If absent, watch the following video to know how to calculate percent composition and ask Ms. Ha for the hamburger activity to complete the worksheet. Below is the notes:
Since students have clear concept on counting atoms, they were introduced to find molar mass of chemical formula. If absent, watch the video, complete the assignment, and turn it in.
Students were introduced how to count atoms in chemical formula. For absent students, watch the following video and complete the worksheet.
Students reviewed over the Unit 5 today. They solved practice questions by rotating stations. Each station had adequate notes and tutorials so that students can learn themselves by annotating the notes. If absent, please review the worksheet and try to solve the questions. Unit 5 exam will be tomorrow (11/11/16).
Students learned how to name and write formula of acids compounds. The attached document contains notes and practice problems. Students who were absent should annotate the notes and solve the practice problems. If you have any questions, please come to the tutorial for help.
Students practiced on the covalent compound naming part.
Students used the covalent foldable to name the compounds.
Students took a quiz over naming compound quiz and made a foldable for covalent compounds
Students learned how to name and write formula of polyatomic ions. They finished the notes and will be using it for their quiz tomorrow. (The note will be only the second page of the document.)
Students learned how to name and write formula of the ionic compounds.
10/26- Students took District Assessment today (multiple choice part only). It will be graded as a Major grade.
10/27- Students took District Assessment writing section (CBD) today for the first 15 minutes. Students reviewed over Unit 1 - Lab safety, Unit 2 - Matter, Unit 3- Periodic Table and Unit 4- Chemical Bonding. Students who were absent: Please complete the review packet and turn it in for a replacement MAJOR grade. (Due Tue. 10/25/16)
a Today we had a class discussion over metallic bond. Students should be able to understand the concept of "Sea of mobile electrons" and know the properties of the metals. Students who were absent: Please watch the following videos and complete the worksheet below.
Students watched a youtube video about Ionic bond and completed the other page of the previous worksheet for another minor grade. For students who were absent: Watch the following video. Also, complete the second page of the bonding worksheet (attached) for a grade. (Students do not have to complete the "molecular geometry" part on the second page.)
Students got 3 different worksheets to complete for extra credits. Students must complete all of the worksheets to get maximum 10 extra credits on a Major grade and a replacement grade of the lowest minor grade.
Exploring ionic covalent bonding is for max. 5 extra credits on major grade.
Covalent compounds worksheet is for max. 5 extra credits on major grade.
HW- E.configuration is for replacement grade for the lowest minor.
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||